Vaxart operates in a single, disruptive segment — the development and commercialization of oral recombinant vaccines administered via temperature-stable tablets.
Vaxart’s proprietary oral vaccine platform, VAAST, aims to improve upon existing vaccine platforms in four ways: mucosal immunity, tolerability, cross-variant protection, and convenience.
Vaxart’s current focus is on developing VXA-CoV2-1, the first oral vaccine candidate for prevention of Covid-19 to be tested in humans in the United States.
Vaxart’s suppressed valuation, originating from a misunderstanding and misrepresentation of Phase I clinical data, provides a unique investment opportunity.
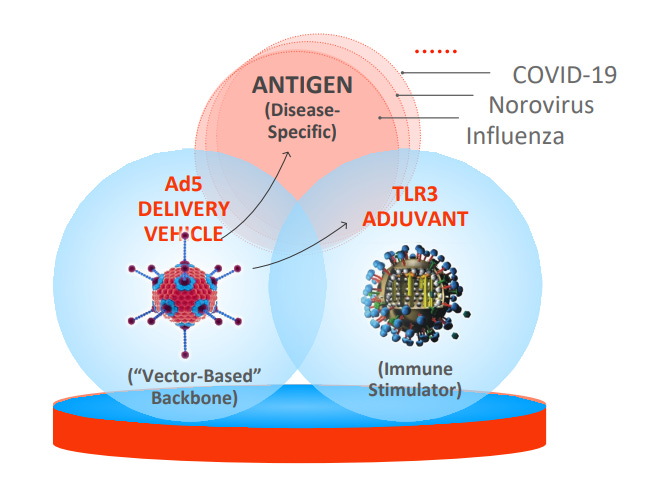
Vector-Adjuvant-Antigen Standardized Technology (VAAST)
Vaxart’s proprietary oral vaccine platform, VAAST, has the potential to transform the vaccine ecosystem. VAAST vaccines are designed to trigger broad immune responses via intestinal delivery. These vaccines have the potential to be more protective than injectable vaccines by activating both systemic and mucosal immunity, preventing infection at points of contact like the nose, lungs, and mouth. By contrast, injectable vaccines only activate systemic immunity and thus do not provide the same protection.
The technology that lies at the heart of VAAST is the adenovirus type 5 virus (Ad5) delivery platform. Ad5 is engineered to be non-infectious and is used as a carrier for the components of the vaccine — the gene coding for “the antigen” and the “adjuvant.” The antigen is a part of the target virus known to stimulate a protective immune response, whereas the TLR3 adjuvant is a molecule that acts to boost the immune response.
Vaxart has completed 13 clinical trials for 7 different viruses and is the only producer of oral vaccines with a platform backed by clinical data against a respiratory pathogen. The results of Vaxart's Phase II trial of their oral flu vaccine (VXA-A1.1) was funded by the U.S. Biomedical Advanced Research and Development Authority (BARDA). This clinical trial compared Vaxart's oral flu vaccine candidate to one of the leading injectable flu vaccines — Sanofi's Fluzone — and found that reduction in illness and infection rates trended superior to Fluzone.
Mucosal Immunity
The same Phase II clinical trial found that the correlate of protection for oral vaccines is very different than for injectables. While Vaxart's oral flu vaccine generated less than one-tenth of the serum neutralizing antibodies of the injectable, it protected just as well.
In order to better understand the antibody immune responses stimulated by VAAST vaccines, one first needs to understand the two primary arms of the immune system and how they function. The immune system fights infections using T cells and B cells. B cells produce various antibodies, including IgA and IgG. IgA antibodies are primarily produced in places such as the GI tract and the respiratory tract, where respiratory viruses like seasonal influenza and SARS-CoV-2 first invade. IgG antibodies are produced in the blood. Traditional injectable vaccines work by inducing a strong IgG response in the blood, whereas oral vaccines induce a strong IgA response in the GI and respiratory tracts, while inducing some IgG responses in the blood as well.
Tolerability
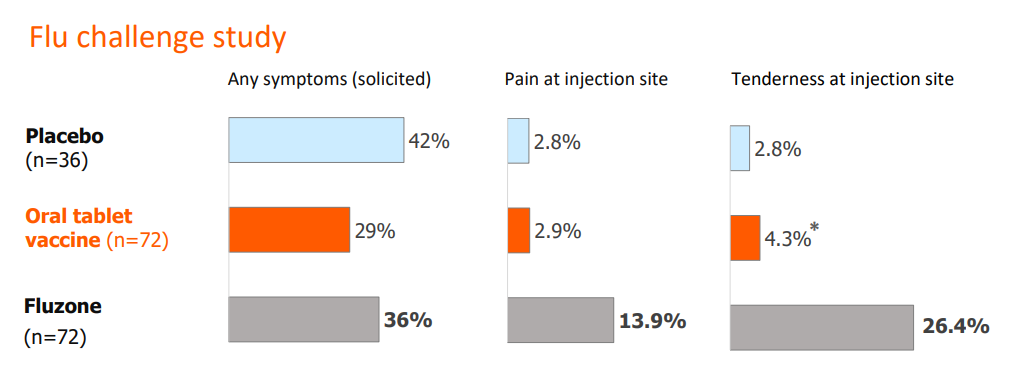
An additional aspect VAAST improves upon with respect to injectable vaccines is tolerability. The potential to experience side effects, as well as an aversion to needles because of the associated pain and lasting tenderness, can act as hurdles to vaccinating a global population. The clinical results comparing a placebo, Fluzone, and VXA-A1.1 are shown below.
VXA-CoV2-1

Vaxart developed their oral solution to Covid-19, VXA-CoV2-1 using VAAST. When the vaccine encounters the immune cells of the lining of the small intestine, they are stimulated to produce an immune response against SARS-CoV-2, the virus that causes the Covid-19 disease.
Preclinical trials studying VXA-CoV2-1 in hamsters showed significant antibody responses against SARS-CoV-2 and prevented symptoms normally seen in infection subjects such as lung damage, inflammation, and lung weight gain, compared to unvaccinated subjects.
On February 3rd, 2021 Vaxart announced preliminary data from its Phase I study in humans to assess safety. Results showed that VXA-CoV2-1 was generally well tolerated. No severe adverse side effects were reported and adverse events were generally mild and gastrointestinal in nature. Including this study, a total of 495 subjects have now been dosed with the VXA-CoV2-1 platform, with no serious adverse events reported.
Preliminary Phase I efficacy data (NCT04563702) showed strong immune responses stimulated by B cells and T cells. The data also revealed that VXA-CoV2-1 resulted in a significant T cell response to both the S (Spike) and N (Nucleocapsid) proteins. This multi-pronged immune response has the potential to provide a more durable, variant resistant protection against SARS-CoV-2.
Phase I efficacy data additionally showed IgA antibodies present in blood and nasal swab samples. Because mucosal IgA can be more potent than serum IgG in neutralizing SARS-CoV-2, it’s possible that the infection could be prevented by even small quantities of IgA in the nose or mouth.
Cross-variant Protection Against COVID-19
One of the most challenging difficulties of solving the Covid-19 problem is the rapid emergence of new variants, especially those with the ability to infect those who survived the original illness with another round of moderate infection. These include the B.1.351 variant originating in South Africa, the B.1.1.7 variant originating in England, and the newly discovered B.1.1.28.1 (lineage P1) which has widely infected the population of Manaus, despite the fact that the city had already experienced widespread infection in May. These new mutations are starting to accumulate in the S1/RBD domain, a subunit of the popularly targeted S protein, where neutralizing antibodies generated by current vaccines bind.
Vaxart’s pill vaccine, VXA-CoV2-1, is among the first Covid-19 vaccine candidates to contain genes for both S and N proteins. The S, or Spike protein acts as a bridge between the virus and the host’s cells that causes infection and the N protein serves as the command center of the SARS-CoV-2 virus itself that both controls the shape and structure of the virus and determines how it replicates. By targeting both proteins, VXA-CoV2-1 is doubling down on targeting the virus’ key building blocks. Additionally, the N protein is more conserved between emerging variants of the virus — its shape is 90% similar to other coronavirus N proteins vs 76% for the S protein — and can provide a long-term target for trying to broaden the responses against all variants.
Vaxart is also advancing S-only second generation vaccine candidates targeted specifically against new variants in which the S protein has mutated. These second generation candidates are designed to complement the potent T cell inducer VXA-CoV2-1 and generate strong mucosal and serum antibody responses. Vaxart has previously shown that a bivalent oral vaccine using its platform can induce immune responses without interference.
As previously mentioned, the highlight of Vaxart’s vaccine is its generation of robust T cell responses post immunization. Vaxart’s vaccine induces a CD8 and CD4 T cell response stronger than any currently approved candidate on the market. This is important not only because T cells are a strong protector against severe infection, but are also generally cross-protective among different variants, holding a broader antigenic repertoire than antibody responses.
Clarifying the Science
Multiple pieces of scientific literature have been published in the past year about the role of antibodies versus T cell response in protective immunity against SARS-CoV-2. In Vaccine, Marc Hellerstein, a professor at UC Berkeley, points out that antibodies are not the primary protective response to infection by coronaviruses. “A strong antibody response correlates with more severe clinical disease in Covid-19, while a strong T cell response is correlated with less severe disease. And antibodies have been short-lived, compared to virus-reactive T cells in recovered SARS patients,” Hellerstein said.
Additionally, T cell responses to SARs-CoV-1 (SARS), a close relative of SARs-CoV-2, have been found to last for 17 years after infection and could potentially be protective against SARs-CoV-2, demonstrating the potential of a robust T cell response to provide durable immunity to a broad range of coronaviruses. Vaxart believes their VXA-CoV2-1 vaccine will be able to provide similarly broad protection.
Although many vaccine developers have focused on antibody production as the primary means to provide protection, there has been little focus on the role of T cells, resulting in a critical blind spot in this area. VXA-CoV2-1 responds to this by marrying the B and T cell response to boost immunity and potentially provide durable efficacy.
Convenience
Not only does VXA-CoV2-1 demonstrate important immunological advantages over other vaccines, an oral Covid-19 vaccine provides key advantages in the area of convenience. We’ll start with the obvious; oral administration is quick and painless and provides an alternative for the estimated 20-30% of the population who have an aversion to needles.
Preparation and administration of injections also require vast resources and can unnecessarily occupy the valuable time of essential healthcare workers who are in short supply. VXA-CoV2-1, and pill vaccines more broadly, are stable at room temperature. This is highly advantageous in regards to the logistics of storage and distribution, which in turn is less taxing on healthcare systems & local resources.
As it stands, thousands of doses a day in the United States and other countries are thrown out or rushed to the nearest available recipient at large vaccination sites, such as sports stadiums and large open venues, with limited access to refrigeration. In countries with more rudimentary healthcare and infrastructure systems, difficulty supplying injectable vaccines only increases.
Instead, governments could be mailing every citizen a vaccine pill for Covid-19, and other viruses to address future outbreaks. Currently, it takes on average of 6+ months to conduct a mass vaccination campaign with injectables. This could be shortened to as little as a few weeks with a pill.
Financials
Accounting for previously reported statistics, Vaxart has a cash and cash equivalent balance of between approximately $180.7 and $192.7 million. With an average historical cash burn of $3 million per month, and $184.2 million in additional funds available via the October 2020 ATM offering, Vaxart has a healthy cash runway and ability to raise additional funds if needed without registering additional shares. While the company saw increased liabilities in 2020, this was primarily due to an increase in development costs and administrative costs associated with nearly quadrupling the size of their staff, and increased legal and consulting fees related to the company’s new focus on the fight against Covid-19.
An important factor to consider that sets Vaxart apart in the biotech sector is that has no debt on its balance sheet. This indicates that the company has been operating purely on its equity investment and has no debt burden. Consensus analysis anticipates the company to incur a final loss in 2021, before generating positive profits in 2022.
Additional Pipeline Highlights
Vaxart is also pursuing several other advances in areas of need aside from Covid-19.
Phase 1 results for Vaxart’s bivalent norovirus vaccine demonstrated IgA ASC response rates of 90 – 93% for GII.4, the most dominant norovirus strain, responsible for over 85% of worldwide outbreaks since the 1990s, and 78 – 86% for GI.1.
[Commonly referred to as the stomach flu], a norovirus vaccine has a commercial opportunity of over $15-20+ billion globally. Vaxart’s norovirus vaccine program was recently restarted with a booster study program for subjects who participated in the Phase Ib trial. Data is expected to be available in the first half of 2021, likely within the second quarter. Additional studies planned for 2021 include a Phase I study in elderly adults and a Phase II challenge study slated for the third quarter.
H1N1 flu vaccine with a completed Phase II efficacy study and an RSV and HPV vaccine in preclinical stages are all on hold pending a licensing, partnering or collaboration agreement or due to the company’s focus on the Covid-19 vaccine.
Universal Flu antigen in collaboration with Janssen Vaccines & Prevention B.V. with an option to negotiate an exclusive license.
Partnerships
Vaxart’s primary collaborative partner is Janssen Vaccines & Prevention B.V., the medical research and product development arm of Johnson & Johnson. Their collaborative efforts are focused on influenza and Vaxart entered into a research collaboration agreement to evaluate the VAAST proprietary oral vaccine platform for the Janssen universal influenza vaccine program. The study was recently completed, and Vaxart has delivered the final report. Janssen has a three-month window, ending May 2021, to negotiate an exclusive license to the technology encompassing the Janssen antigens.
In November 2020, Vaxart also confirmed an expanded collaboration with Kindred Biosciences for the manufacturing of the Covid-19 oral vaccine, and a master services agreement with Attwill Vascular Technologies, LP for processing, lyophilizing, and tableting compounds for the oral Covid-19 vaccine.
Competition
Given the difficulties involved in storing and distributing most Covid-19 vaccines, Vaxart is not the only vaccine producer interested in the prospect of an oral alternative. Competing vaccine developers of note include Oramed Pharmaceuticals and Symvivo Corporation.
Oramed Pharmaceuticals is a joint venture between Oravax Medical and Premas Biotech. By leveraging Oramed’s proprietary oral delivery technology and Premas Biotech’s vaccine technology, Oramed intends to develop and commercialize their own oral Covid-19 vaccine. Initial clinical trials are slated for Q2 of 2021.
Symvivo Corporation is a private company backed by Merck with a Covid-19 vaccine currently in Phase 1 trials. Their candidate makes use of their proprietary BacTRL-Spike infectious disease platform.
Both competing vaccines are in early stages of clinical trials and Vaxart has a clearly defined lead in comparison.
A couple other names that are occasionally mentioned in the oral Covid-19 space are Ridgeback Pharmaceuticals, backed by Merck, and Pfizer. Both are developing oral Covid-19 therapies. Neither are direct competitors to Vaxart’s VXA-CoV2-1 vaccine as these antiviral treatments are focused on treating patients who already have already been infected with Covid-19.
Investment Thesis
Much noise has been made in the investment community about the recent Phase I trial results for Vaxart’s Covid-19 vaccine showing low neutralizing antibody responses. The press compared these results to the current wave of first generation vaccines. Our fundamental thesis anticipates that these results have been broadly misunderstood and even misrepresented. The metrics governing success in Vaxart’s vaccine cannot be directly compared to its current peers.
Although neutralizing antibodies are necessary overall, the amount we see produced in the current crop of injectable vaccines is unnecessary for Vaxart’s oral vaccine. As mentioned above, Vaxart’s oral flu vaccine candidate was more effective with less than one-tenth of the neutralizing antibodies as Fluzone. The case is likely the same for their Covid-19 vaccine.
If Vaxart succeeds in developing their oral Covid-19 vaccine either as a primary second generation vaccine or as a booster for those already vaccinated or exposed to the virus, the total addressable market is considerable both in the United States and worldwide. As it stands, the ability to cope with current and future pandemics is greatly limited by the ability to store, transport and distribute vaccines effectively. Vaxart, with their proprietary platform, VAAST, is a notable innovator in this space. As a first mover and industry leader, Vaxart could potentially become a principal source of the global vaccine supply.
Taking into account their proprietary technology, cash flow, total addressable market for their products and lastly historical price action; we rate Vaxart as a speculative BUY and set a 2021 preliminary price target of $22.
Risks
We have identified some risk factors that could stand in the way of our investment thesis.
There does exist the possibility that Vaxart’s VXA-CoV2-1 vaccine does not achieve statistically significant enough protection to warrant FDA approval. While always a risk with discovery and development stage biotech companies, the risk here is slightly more pronounced. Although Vaxart’s Covid-19 vaccine does not require a strong neutralizing antibody response, it still needs some response, and their Phase I trial had instances of a total lack of neutralizing antibody response with certain dosing regimens. Should Vaxart be unable to find a proper dosing regimen that generates sufficient antibody responses, Vaxart’s development schedule could stall, dealing a major blow to the company.
That being said, based on the data available, there is a significant possibility that even with a total lack of IgG antibody response, Vaxart’s vaccine could be used as a prophylaxis providing short-term immunity against Covid-19 that could last up to a month at a time. Even this indication would likely allow them to recover a significant portion of their R&D investment in the vaccine.
The second major risk factor is that Vaxart’s current oral vaccine arrives too late to become a major player in this space. As it stands, their Covid-19 vaccine trial is entering its second phase in Q2 2021. An optimistic estimate would place the likelihood of a potential approval somewhere in Q4 2021, a time by which we hope to have a significant portion of the world vaccinated against Covid-19.
However, with vaccine skepticism at an all-time high and SARS-CoV-2’s ability to easily jump hosts and mutate, many experts believe that Covid-19 will become an endemic disease that stays circulating among the populace for an extended period,. Achieving herd immunity may be a years-long initiative.
Vaxart directly addressed this particular concern in their most recent investor presentation. Because VAAST vaccines are likely to be well-tolerated as a supplement to the current wave of vaccines, they consider their role in this pandemic to develop vaccines that will serve as a yearly booster to the others and with multiple versions produced as a way to react quickly to new variants resistant to the current wave of vaccines. Vaxart’s executives have mentioned that getting approved as a booster is far less time-intensive and expensive and they already have plans in place to do just that, with their next wave of studies being planned at a variety of locations worldwide to test against several prominent regional variants.
Final Thoughts
Keeping all of the aforementioned risks in mind, while a total failure of their Covid-19 vaccine candidate would be a major setback, it would not spell the end for Vaxart as a company or even the end for Vaxart’s ability to reach its full potential in the long-term.
Their norovirus and seasonal flu vaccine candidates already show promising results. Success in either of these ventures would translate to success of Vaxart as a company. Sanofi’s Fluzone generates an average $1 billion per quarter and a norovirus vaccine is estimated to have a $15-20+ billion total addressable market globally — $10+ billion of which is driven by United States government policy. Either of these revenue pipelines could justify a multi-billion dollar market capitalization for Vaxart.
Looking ahead, we anticipate current developments as merely the beginning of the company’s story. Similarly to how Moderna’s RNA vaccine became a vehicle to catalyze the success of the company’s RNA technology, we project Vaxart’s Covid-19 vaccine as a vehicle to jumpstart the success of VAAST oral vaccine platform.
At that juncture, funding for their broader pipeline and licensing interest in the VAAST platform would make Vaxart a revolutionary player in the vaccine space, and likely lead to a valuation that eclipses our preliminary price target.
Disclosure
I am/we are long shares and 2023 LEAPS of VXRT. This article was written by our independent team and expresses our own opinions. We are not receiving compensation and have no business relationship with any company whose stock is mentioned in this article.
Sources and Further Reading
Vaxart’s Oral Vaccine Candidate for Prevention of Covid-19: Link
Developing the Pill Against Covid-19: Link
Fast Facts: Link
Form 8-K 2021-02-25: Link
Form 8-K 2021-02-03: Link
What are the roles of antibodies versus a durable, high quality T-cell response in protective immunity against SARS-CoV-2?: Link